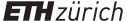Intended learning outcomes: Describe flow resources within a process stage. Explain the need for flexible capability (of the production equipment) in chemical production.
The following values of characteristic features indicate processor-oriented methods as the appropriate business methods for planning & control:
Production environment: In the process industry, end products stores, and thus make-to-stock are widespread and important. Chemical, pharmaceutical, or grocery products are, ultimately, stocked at the shelf in retail shops. Upstream added value stages are also kept in stock, where efficient.
Facility layout: Here, we find high-volume line production, and — in particular — continuous production. Production processes in process industries (producing chemicals, paint, oil, and so on) usually have to carry out an entire sequence of operations (a process stage; see the definition below), that is, one operation after another in a continuous fashion.
A flow resource F is an intermediate product that should not or cannot be stored during the process stage and therefore flows through the process continuously.
An intermediate product becomes a flow resource mainly because of its physical nature or condition. An example is the active substances produced in the chemical industry. As a data element in the product structure, a flow resource is at the same level as the component materials for the subsequent operation or (basic) manufacturing step, and it facilitates modeling and monitoring of the balance of material inputs and outputs of individual manufacturing steps. Figure 8.1.2.1 shows a product Z produced from starting material G.
Fig. 8.1.2.1 Flow resources within a process stage.
The intermediate states F1 and F2 “flow,” meaning that they are not, or cannot, be stored in containers or tanks. Thus, they cannot be in buffers at these work centers[note 801]. This also means that storable work in process cannot build up at these work centers. This reduces the degree of freedom for capacity planning (that leeway is utilized in the conventional MRP II concept; see the comments on queues in Section 13.2).
Example: Flow Resource
In practice, flow resources usually refers to fluids, gases or bulk goods that are moved forward between work centers and are not stored in-between, or to materials that cannot be stored, depending on the circumstances. From the point of view of planning and control, one result is that the stations connected to each other by the flow resources have to be considered as one unit. Thus, there is no freedom for individual capacity planning of each work center. The effects of even a temporary lapse in attention is shown in the following Flash animation in a drastic but thoroughly realistic way.
Flexible capability (of the production equipment): Single-purpose facilities, i.e. dedicated capacity, [note 806] were common in chemical production for a long time. For very large-scale mass production, there are sound economic reasons for this type of structure. However, to adapt capacity to load more flexibly and particularly to facilitate change of product on the same production resources, multipurpose facilities composed of modules became more frequent. Nevertheless, they are far away from achieving the flexibility of mechanical production. Inflexible facilities still exist, not least because of conditions imposed by government regulations. See also [Hübe96], p. 23 ff. Food and drug production are subject to strict quality control by bodies such as the FDA (U.S. Food and Drug Administration). Production of foodstuffs and drugs must follow a set of guidelines known as Good Manufacturing Practices, or GMP (also known as Quality System Regulation). Under GMP, manufacturing practices are inspected and approved at each plant, which means that it is not possible to simply switch production between facilities in response to temporary capacity shortages or mechanical faults, for example. The production process would also have to be validated at the alternative facility.
Course section 8.1: Subsections and their intended learning outcomes

8.1 Characteristics of the Process Industry
Intended learning outcomes: Explain divergent product structures and by-products. Describe high-volume line production, flow resources and inflexible facilities. Produce an overview on large batches, lot traceability, and loops in the order structure.

8.1.1 Divergent Product Structures, Primary Products, By-Products, and Waste Products
Intended learning outcomes: Differentiate between primary product, by-product, and waste product. Explain the manufacture of by-products in chemical production.

8.1.1b Manufacture of By-Products in Mechanical Production: Sheet Metal Working
Intended learning outcomes: Describe the manufacture of by-products in sheet metal working.

8.1.1c Manufacture of By-Products in Mechanical Production: Collets from a Steel Cylinder and Temporary Assembly
Intended learning outcomes: Disclose the production of collets from a steel cylinder as well as the “saucepan and lid” problem linked with temporary assembly.

8.1.2 High-Volume Line Production, Flow Resources, and Inflexible Facilities
Intended learning outcomes: Describe flow resources within a process stage. Explain the need for flexible capability (of the production equipment) in chemical production.

8.1.3 Large Batch Size, Lot Traceability, and Loops in the Order Structure
Intended learning outcomes: Explain the need for lot traceability. Present reasons for loops in the product structure.